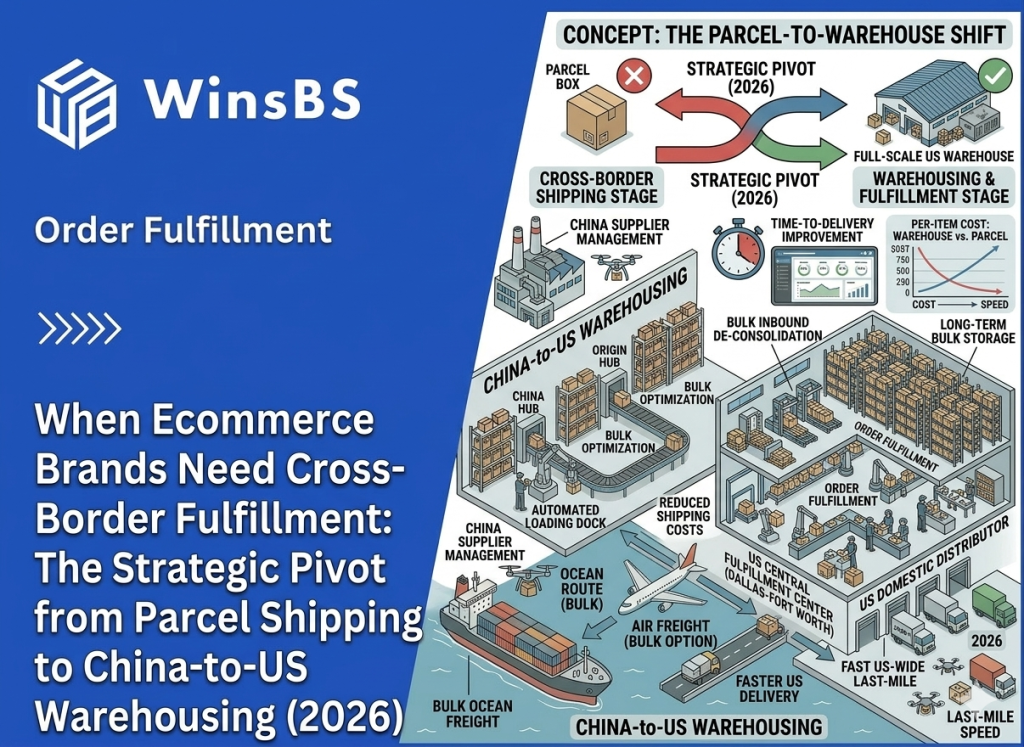
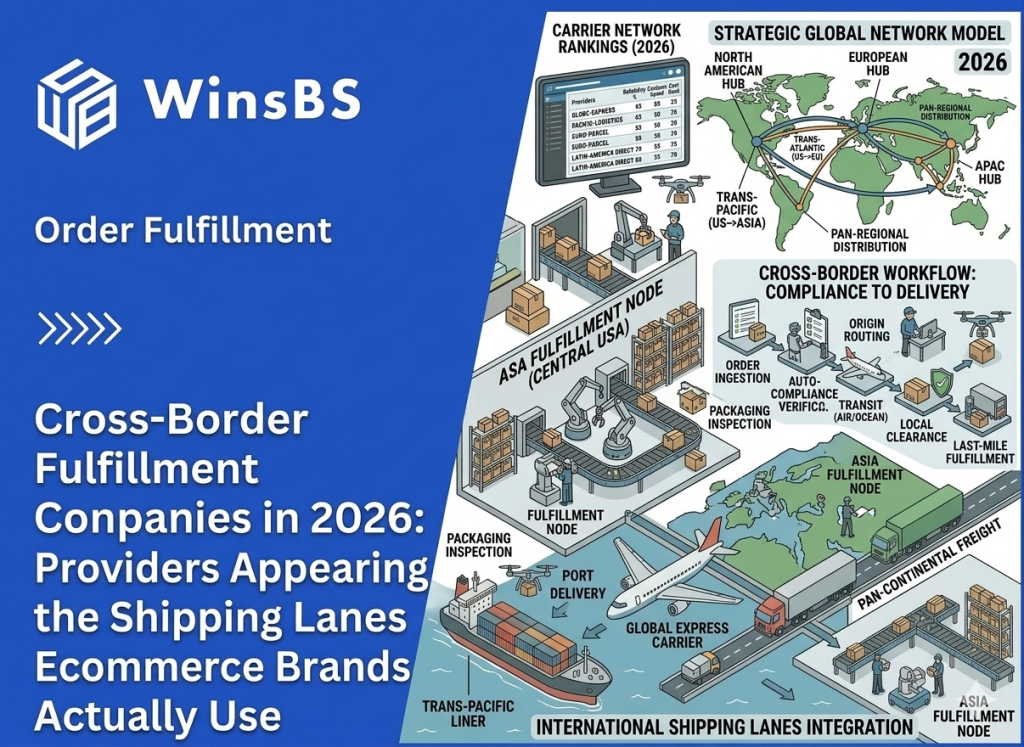
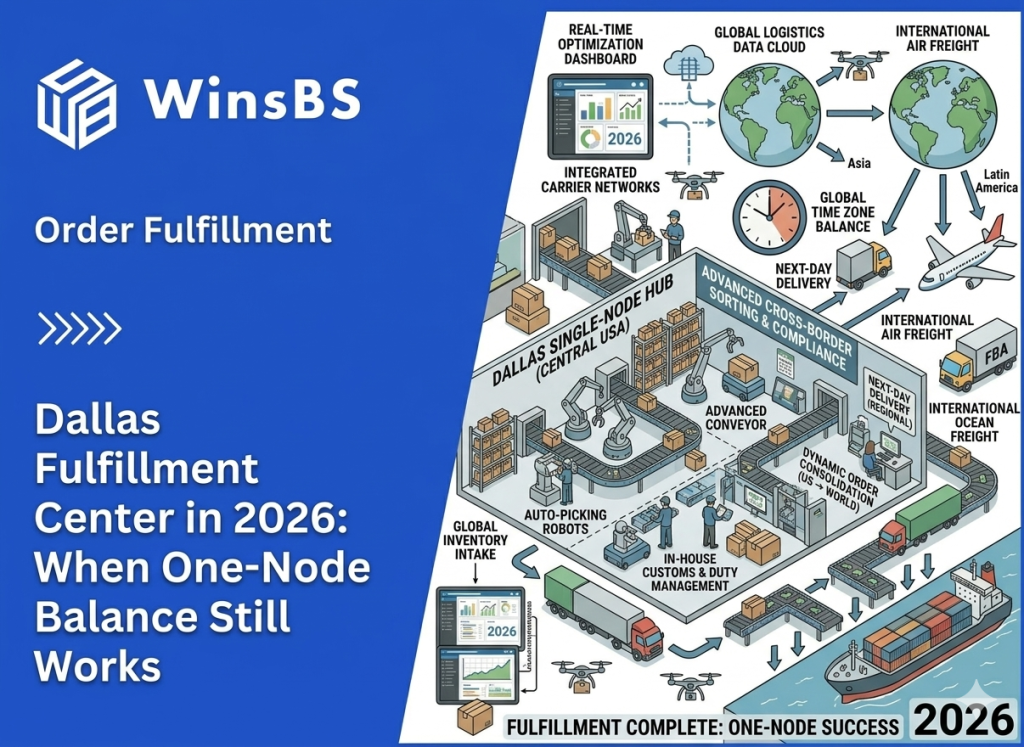
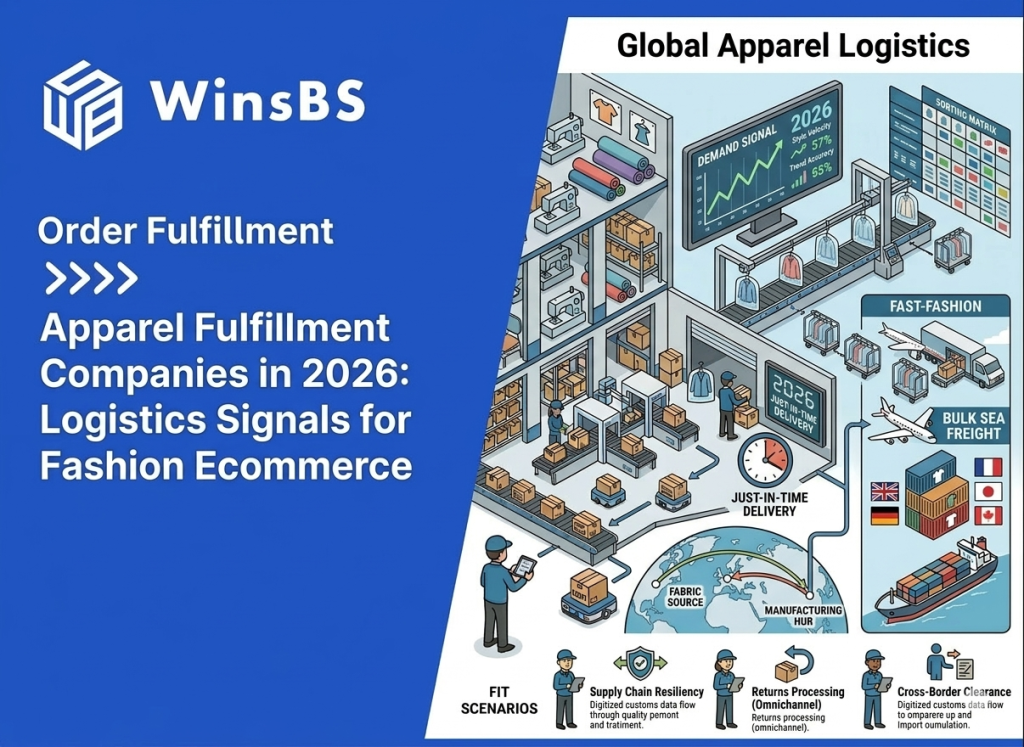
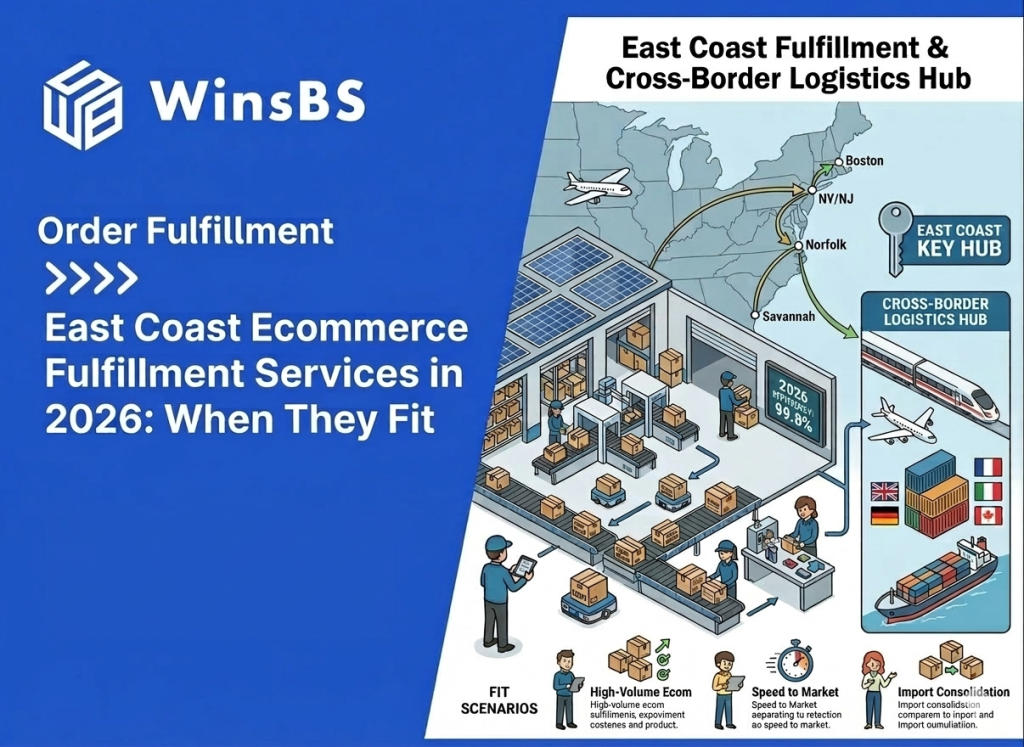
When Ecommerce Brands Need Cross-Border Fulfillment: The Strategic Pivot from Parcel Shipping to China-to-US Warehousing (2026)
When Ecommerce Brands Need Cross-Border Fulfillment The Strategic Pivot from Parcel Shipping to Inventory Positioning WinsBS Fulfillment ORDER FULFILLMENT COMPANY · MAXWELL ANDERSON March 2026 Quick Context Many ecommerce brands do not begin with cross-border fulfillment. They begin with direct parcel shipping from China because it is simple, flexible, and workable at small scale. The pivot usually comes later, when delivery visibility weakens, shipping costs become less predictable, and customer experience starts depending on a logistics structure that was never designed for stable international growth. This article explains when that shift happens and why the real decision is no longer about choosing a faster parcel service, but about repositioning inventory closer to the destination market. Table of Contents Why Direct Parcel Shipping Works in the Early Stage Why Shipping Friction Increases as Volume Grows How Cross-Border Logistics Systems Become Complex Where the Parcel Shipping Model Reaches Its Limits Why Inventory Positioning Changes Delivery Stability The Typical China–US Cross-Border Fulfillment Structure When the Operational Pivot Point Has Already Arrived Cross-Border Fulfillment as a Way to Rebuild Delivery Control For readers comparing this article with broader provider-focused research, the logistics logic here sits one level below a company list. The question is not which cross-border fulfillment providers exist, but why ecommerce brands that begin with direct parcel shipping eventually reach a point where inventory positioning, delivery control, and route stability matter more than parcel convenience alone. Why Direct Parcel Shipping Works in the Early Stage For many ecommerce brands manufacturing in China, direct parcel shipping is the most practical way to begin selling internationally. When orders start arriving from overseas customers, sending individual parcels directly from the origin warehouse allows brands to reach global buyers without building additional infrastructure. In its simplest form, the logistics path is straightforward. Products leave a warehouse in China, move through an international parcel carrier, pass through destination-country customs, and are then transferred to a local delivery network for final distribution. This structure is widely used across cross-border ecommerce and is supported by international shipping programs offered by carriers such as USPS international shipping services and other global parcel networks. At a small scale, the model works because it keeps operational requirements minimal. Brands can ship each order as it is placed, avoiding the need to position inventory in another country before demand becomes predictable. There is no overseas warehouse to manage, no inventory forecasting tied to a specific market, and no commitment to a single distribution hub. This flexibility is particularly valuable in the early stages of international growth. Demand may come from several regions at once, and the brand may not yet know which market will develop consistent order volume. Direct parcel shipping allows products to reach customers in multiple countries while the business is still learning where its strongest demand actually exists. Another reason the model works early on is that operational complexity remains manageable. When daily order volume is relatively low, handling each shipment individually does not create significant pressure on logistics coordination, customer service, or carrier performance. Shipping costs are predictable enough, and delivery variability usually remains within acceptable limits for customers who are already expecting international transit times. Industry documentation around international parcel services reflects this reality. Global postal networks and parcel carriers have long developed cross-border ecommerce delivery programs specifically designed for low-to-moderate shipping volumes, a structure widely documented by organizations such as the Universal Postal Union’s ecommerce logistics research. In other words, direct parcel shipping is not an inefficient logistics strategy. In the right conditions, it is the most logical one. It allows brands to test international demand, operate with minimal infrastructure, and keep logistics decisions flexible while the business is still defining its market footprint. The challenge appears later, when the same conditions that once made parcel shipping practical begin to change. As international orders concentrate in specific markets and shipping volumes grow — a trend documented in global cross-border ecommerce research such as Statista’s cross-border ecommerce analysis — the logistics structure that once provided flexibility can gradually become a source of instability. Understanding how that shift happens is the key to recognizing when parcel-based logistics begins reaching its limits. Why Shipping Friction Increases as Volume Grows As international order volume increases, many ecommerce brands begin to notice subtle changes in delivery performance. Shipments that once moved through the logistics chain with relatively predictable timing start showing wider delivery windows, occasional tracking gaps, or unexpected pauses during international transit. In the early stage of cross-border growth, these irregularities are often dismissed as isolated shipping incidents. A parcel may remain in transit for several days without a tracking update, or appear to stall briefly before reappearing in the destination country’s delivery network. Because these situations occur sporadically, they are usually treated as individual carrier issues rather than signs of a structural change. However, when shipping volume continues to rise, the pattern gradually becomes more visible. Customer service teams begin receiving more questions about delivery status, more requests for tracking clarification, and more messages from customers unsure whether their order is still moving through the system. In many cases, the brand itself has limited visibility into the shipment’s exact location during these periods. This shift does not necessarily mean that logistics providers are performing worse. Instead, it reflects the structure of international parcel delivery itself. A direct cross-border shipment typically passes through several independent operational systems before it reaches the final customer. A typical path may begin at a warehouse in China, move into an export carrier network, travel through international transportation routes, pass inspection or processing within destination-country customs, and finally transfer into a domestic delivery network. Each stage is managed by a different organization operating under its own infrastructure and regulatory environment. When parcel volumes remain small, these transitions between systems rarely create noticeable disruption. But as the number of shipments increases, the probability of delays, inspection variations, routing adjustments, or temporary tracking inconsistencies naturally grows. What once appeared to be a simple logistics path begins to